Home / Training / Manuals / Colposcopy and treatment of cervical intraepithelial neoplasia: a beginners manual / Chapter 13: Treatment of Cervical Intraepithelial Neoplasia by Loop Electrosurgical Excision Procedure (LEEP)
 table 13.1: The eligibiligy criter...
table 13.1: The eligibiligy criter... table 11.1: Treatment for reproduc...
table 11.1: Treatment for reproduc... figure 13.3: Instrument tray for L...
figure 13.3: Instrument tray for L... figure 4.9: Vaginal speculum cover...
figure 4.9: Vaginal speculum cover...
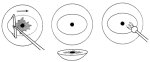 figure 13.4: Excision of an ectoce...
figure 13.4: Excision of an ectoce... figure 13.5: Excision of an ectoce...
figure 13.5: Excision of an ectoce...
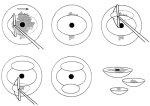 figure 13.6: Excision of an ectoce...
figure 13.6: Excision of an ectoce... figure 13.3: Instrument tray for L...
figure 13.3: Instrument tray for L...
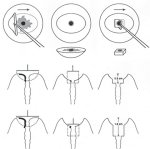 figure 13.7: Excision of ectocervi...
figure 13.7: Excision of ectocervi...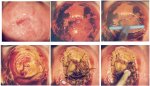 figure 13.8: Excision of an ectoce...
figure 13.8: Excision of an ectoce... figure 13.4: Excision of an ectoce...
figure 13.4: Excision of an ectoce... figure 13.5: Excision of an ectoce...
figure 13.5: Excision of an ectoce... figure 13.6: Excision of an ectoce...
figure 13.6: Excision of an ectoce... FIGURE 13.8: Excision of an ectoce...
FIGURE 13.8: Excision of an ectoce...
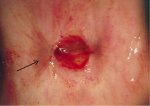 figure 13.9: Appearance of the cer...
figure 13.9: Appearance of the cer...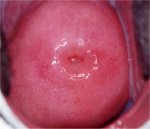 figure 13.10: Appearance of the ce...
figure 13.10: Appearance of the ce...
 table 11.1: Treatment for reproduc...
table 11.1: Treatment for reproduc...
Colposcopy and treatment of cervical intraepithelial neoplasia: a beginners manual, Edited by J.W. Sellors and R. Sankaranarayanan
Chapter 13: Treatment of Cervical Intraepithelial Neoplasia by Loop Electrosurgical Excision Procedure (LEEP)
Filter by language: English / Français / Español / Portugues / 中文- Electrosurgical current applied to tissues can have one of three effects on the tissue, depending on the power setting and the waveform of the current used: desiccation, cutting, and fulguration.
- Loop electrosurgical excision procedure (LEEP) is a relatively simple procedure that can be readily learnt.
- The key advantage of LEEP over cryotherapy is that it removes rather than destroying the affected epithelium, allowing histological examination of the excised tissue.
- A loop wider than the lesion(s) and the transformation zone to be removed should be used; otherwise, the lesion should be removed with multiple passes.
- If the lesion involves the endocervical canal, a two-layer excisional method should be used.
- Women will have a brown or black discharge for up to two weeks after LEEP.
- Women should be advised not to use a vaginal douche, tampon, or have sexual intercourse for one month after LEEP.
- Moderate to severe post-operative bleeding occurs in less than 2% of treated women and they should be seen promptly.
- The failure rate with LEEP in women treated for the first time is around 10%.
Electrosurgery is the use of radiofrequency electric current to cut tissue or achieve haemostasis. A loop electrosurgical excision procedure (LEEP) operator needs to keep in mind that electricity flows to ground along the path of the least electrical resistance. The electrical energy used in electrosurgery is transformed into heat and light energy. The heat from a high-voltage electrical arc between the operating electrode and tissue allows the practitioner to cut by vaporizing tissue (at 100oC) or to coagulate by dehydrating tissue (above 100oC). The cutting electrodes are loops of very fine (0.2 mm) stainless steel or tungsten wire to achieve different widths, depths, and configurations of cut (Figure 13.1).
The higher temperatures involved in coagulation produce thermal effects greater than in electro-surgical cutting. This is important in electrosurgery, since an adequate pathological examination requires that the coagulation effect be minimal in the excised surgical specimen. On the other hand, some coagulation effect is desirable, even while cutting, in order to minimize bleeding in the surgical field. Manufacturers of modern electrosurgical generators (Figure 13.2) are aware of the need to control bleeding. They offer electrosurgical cutting settings that lead to some coagulation by blending electrical currents, one with a cutting waveform and another with a coagulation waveform. This combination is called a blended cutting waveform, and is the type of waveform that will be referred to in this manual when electrosurgical cutting is discussed.
When the coagulation setting is selected on the electrosurgical generator, the coagulation waveform has a higher peak-to-peak voltage (producing higher temperatures) than that used for a pure cutting waveform, and is meant only to heat the tissue above 100°C to achieve dehydration. There are three types of coagulation: desiccation, in which the active electrode touches the tissue; fulguration, in which the active electrode does not touch the tissue but sprays multiple sparks between itself and the tissue; and puncture coagulation, in which an electrode, usually a needle, is inserted into the centre of a lesion. Coagulation using the fulguration setting and a 3- to 5-mm ball electrode is the type of coagulation that is normally referred to in this manual (one exception is be the use of a needle electrode to fulgurate a stubborn area of bleeding). The fulguration setting uses a higher peak-to-peak voltage waveform than the other coagulation settings, coagulating tissue with less current and, therefore, less potential harm to adjacent tissue.
To obtain a proper effect, an electrosurgical generator requires that a patient-return electrode or dispersive plate be used to allow the electrical circuit to be completed and the optimal current to flow. The dispersive plate should always be placed as close to the surgical site as possible. This is in contrast to the desired effect at the active electrode, where the current density is purposely high to concentrate the electrical energy as it is transformed into heat. Unless good electrical contact is maintained at the dispersive electrode over a large area, there is a danger that the patient will suffer from an electrical burn at this site. To guard against this possibility, modern electrosurgical units have circuitry (commonly referred to as a return electrode monitoring system) that continuously monitors the adequacy of the ground plate (dispersive pad) connection to the patient. This type of circuitry not only alerts the operator of a problem, but also prevents operation until the circuit fault is corrected. It is highly recommended that any electrosurgical generator meet the basic standards described above to ensure that safe and effective electrosurgery can be performed. It is assumed that only a system that meets or exceeds such requirements will be used in any of the electrosurgical procedures described in this manual.
Electrosurgery must not be performed in the presence of flammable gases, flammable anaesthetics, flammable liquids (e.g., alcohol-containing skin-preparation solutions or tinctures), flammable objects, oxidizing agents, or an oxygen-enriched atmosphere. The operator is, of course, at risk of receiving a burn from the active electrode if it is accidentally touched while activated.
The higher temperatures involved in coagulation produce thermal effects greater than in electro-surgical cutting. This is important in electrosurgery, since an adequate pathological examination requires that the coagulation effect be minimal in the excised surgical specimen. On the other hand, some coagulation effect is desirable, even while cutting, in order to minimize bleeding in the surgical field. Manufacturers of modern electrosurgical generators (Figure 13.2) are aware of the need to control bleeding. They offer electrosurgical cutting settings that lead to some coagulation by blending electrical currents, one with a cutting waveform and another with a coagulation waveform. This combination is called a blended cutting waveform, and is the type of waveform that will be referred to in this manual when electrosurgical cutting is discussed.
When the coagulation setting is selected on the electrosurgical generator, the coagulation waveform has a higher peak-to-peak voltage (producing higher temperatures) than that used for a pure cutting waveform, and is meant only to heat the tissue above 100°C to achieve dehydration. There are three types of coagulation: desiccation, in which the active electrode touches the tissue; fulguration, in which the active electrode does not touch the tissue but sprays multiple sparks between itself and the tissue; and puncture coagulation, in which an electrode, usually a needle, is inserted into the centre of a lesion. Coagulation using the fulguration setting and a 3- to 5-mm ball electrode is the type of coagulation that is normally referred to in this manual (one exception is be the use of a needle electrode to fulgurate a stubborn area of bleeding). The fulguration setting uses a higher peak-to-peak voltage waveform than the other coagulation settings, coagulating tissue with less current and, therefore, less potential harm to adjacent tissue.
To obtain a proper effect, an electrosurgical generator requires that a patient-return electrode or dispersive plate be used to allow the electrical circuit to be completed and the optimal current to flow. The dispersive plate should always be placed as close to the surgical site as possible. This is in contrast to the desired effect at the active electrode, where the current density is purposely high to concentrate the electrical energy as it is transformed into heat. Unless good electrical contact is maintained at the dispersive electrode over a large area, there is a danger that the patient will suffer from an electrical burn at this site. To guard against this possibility, modern electrosurgical units have circuitry (commonly referred to as a return electrode monitoring system) that continuously monitors the adequacy of the ground plate (dispersive pad) connection to the patient. This type of circuitry not only alerts the operator of a problem, but also prevents operation until the circuit fault is corrected. It is highly recommended that any electrosurgical generator meet the basic standards described above to ensure that safe and effective electrosurgery can be performed. It is assumed that only a system that meets or exceeds such requirements will be used in any of the electrosurgical procedures described in this manual.
Electrosurgery must not be performed in the presence of flammable gases, flammable anaesthetics, flammable liquids (e.g., alcohol-containing skin-preparation solutions or tinctures), flammable objects, oxidizing agents, or an oxygen-enriched atmosphere. The operator is, of course, at risk of receiving a burn from the active electrode if it is accidentally touched while activated.
Practising LEEP and demonstrating competence before use on patients
It is mandatory that every colposcopist has practised and demonstrated the ability to perform LEEP adequately by simulating the excision of cervical lesions on meat (beef, pork etc.) or fruits on which mock lesions have been painted to scale. Typewriter correction fluid or trichloroacetic acid work well for painting mock lesions. LEEP should always be practised using the colposcope, as is done in actual practice. If possible, colposcopists should have experience and demonstrated competence with cryotherapy before learning LEEP.
The step-by-step approach to LEEP
First, it must be confirmed that the woman meets the eligibility criteria in Table 13.1.
If there is evidence of pelvic inflammatory disease (PID), cervicitis, vaginal trichomoniasis, bacterial vaginosis or anogenital ulcer, it is advisable to delay LEEP until that condition has been treated and resolved (see Chapter 11, Table 11.1). If there is marked atrophy due to estrogen deficiency in an older woman and staining of the outer margin of a lesion is indistinct, it is advisable to delay LEEP until after a course of topical estrogen treatment.
It is generally preferable to have the diagnosis of CIN firmly established before LEEP is performed. However, there may be exceptions to this general rule, for example, in the context of developing country settings, women may be offered treatment at their first colposcopy visit to maximize treatment coverage (otherwise patients lost to follow-up would not receive treatment for lesions). Expert colposcopists also may use this approach to maximize treatment coverage and to minimise the number of clinic visits in some clinical settings.
The instruments needed for LEEP should be placed on an instrument trolley or tray (Figure 13.3). If the woman is returning to the clinic on a second visit for treatment, colposcopic assessment should be carried out immediately before LEEP to confirm that the location and linear extent of the lesion are amenable to effective LEEP. The application of Lugols iodine solution is helpful to outline lesion margins before the start of treatment. An insulated vaginal speculum (Figure 13.3) with an electrically insulating coating or a speculum covered with a latex condom (Figure 4.9) should be used to avoid an electrical shock to the woman in the event that the activated electrode inadvertently touches the speculum (though this type of event usually does not cause any tissue damage because of the relatively large area of contact). Similarly, care must be taken to avoid causing pain by inadvertently touching the vaginal walls with the activated electrode. The later possibility may be avoided by using an insulated vaginal sidewall retractor in addition to an insulated vaginal speculum (Figure 13.3) or by using a speculum covered by a condom (Figure 4.9).
It is ideal if the vaginal speculum used has a smoke evacuator tube attached to the luminal surface of the anterior blade so that a source of suction can be attached. If this type of speculum is not available, a simple suction tube (preferably made of non-conductive and non-flammable material) may be used, and the open tip should be positioned as near as possible to the cervix. A smoke evacuation system with a high rate of flow and a means of filtering out the smoke particles and odour is mandatory.
Local anaesthesia is achieved 30 seconds after multiple injections of a total of 5ml or less of 1% xylocaine (or a similar agent) into the stromal tissue of the ectocervix. The injections are given in a ring pattern 1 -2 mm deep (at 3, 6, 9 and 12 oclock positions) at the periphery of the lesion and transformation zone using a 5ml syringe and 25- to 27-gauge needle. It is common practice to reduce the amount of bleeding during the procedure by mixing a vasoconstrictor agent such as vasopressin (no more than one pressor unit) with the injected local anaesthetic agent. The use of xylocaine with 2% adrenaline instead of pitressin also is adequate for local anaesthesia, but may cause palpitations and leg tremors before surgery. However, this can be avoided if infiltration is subepithelial. If a two-layer excision (LEEP cone) is planned, local anaesthetic is injected into the anterior and posterior endocervical canal also.
The aim of the LEEP procedure is to remove the lesions and the transformation zone in their entirety and send the affected tissue to the histopathological laboratory for examination. The least amount of power that will effectively perform the electrosurgery should be used, so as to minimize the risk to the patients normal tissues and ensure that the excised specimen is in acceptable condition (with a minimum of thermal artifact) for pathological assessment. The power setting used depends on the size of the tissue electrode being used for cutting and whether fulguration is being performed - this information should be predetermined in each clinic and be available to the LEEP operator when choosing a power setting on the machine for cutting or fulguration. The commonly used power settings for the different loop electrodes are as follows: 1.0 ´ 1.0 cm 30 watts; 1.5 ´ 0.5 cm 35 watts; 2.0 ´ 0.8 cm 40 watts; 2.0 ´ 1.2 cm 50 watts. The power settings for 3 mm and 5 mm ball electrodes are 30 watts and 50 watts, respectively, in the coagulation mode. When possible, a lesion should be removed with one pass of the loop electrode, although this is not always feasible. Four basic operative scenarios are described below:
It is generally preferable to have the diagnosis of CIN firmly established before LEEP is performed. However, there may be exceptions to this general rule, for example, in the context of developing country settings, women may be offered treatment at their first colposcopy visit to maximize treatment coverage (otherwise patients lost to follow-up would not receive treatment for lesions). Expert colposcopists also may use this approach to maximize treatment coverage and to minimise the number of clinic visits in some clinical settings.
The instruments needed for LEEP should be placed on an instrument trolley or tray (Figure 13.3). If the woman is returning to the clinic on a second visit for treatment, colposcopic assessment should be carried out immediately before LEEP to confirm that the location and linear extent of the lesion are amenable to effective LEEP. The application of Lugols iodine solution is helpful to outline lesion margins before the start of treatment. An insulated vaginal speculum (Figure 13.3) with an electrically insulating coating or a speculum covered with a latex condom (Figure 4.9) should be used to avoid an electrical shock to the woman in the event that the activated electrode inadvertently touches the speculum (though this type of event usually does not cause any tissue damage because of the relatively large area of contact). Similarly, care must be taken to avoid causing pain by inadvertently touching the vaginal walls with the activated electrode. The later possibility may be avoided by using an insulated vaginal sidewall retractor in addition to an insulated vaginal speculum (Figure 13.3) or by using a speculum covered by a condom (Figure 4.9).
It is ideal if the vaginal speculum used has a smoke evacuator tube attached to the luminal surface of the anterior blade so that a source of suction can be attached. If this type of speculum is not available, a simple suction tube (preferably made of non-conductive and non-flammable material) may be used, and the open tip should be positioned as near as possible to the cervix. A smoke evacuation system with a high rate of flow and a means of filtering out the smoke particles and odour is mandatory.
Local anaesthesia is achieved 30 seconds after multiple injections of a total of 5ml or less of 1% xylocaine (or a similar agent) into the stromal tissue of the ectocervix. The injections are given in a ring pattern 1 -2 mm deep (at 3, 6, 9 and 12 oclock positions) at the periphery of the lesion and transformation zone using a 5ml syringe and 25- to 27-gauge needle. It is common practice to reduce the amount of bleeding during the procedure by mixing a vasoconstrictor agent such as vasopressin (no more than one pressor unit) with the injected local anaesthetic agent. The use of xylocaine with 2% adrenaline instead of pitressin also is adequate for local anaesthesia, but may cause palpitations and leg tremors before surgery. However, this can be avoided if infiltration is subepithelial. If a two-layer excision (LEEP cone) is planned, local anaesthetic is injected into the anterior and posterior endocervical canal also.
The aim of the LEEP procedure is to remove the lesions and the transformation zone in their entirety and send the affected tissue to the histopathological laboratory for examination. The least amount of power that will effectively perform the electrosurgery should be used, so as to minimize the risk to the patients normal tissues and ensure that the excised specimen is in acceptable condition (with a minimum of thermal artifact) for pathological assessment. The power setting used depends on the size of the tissue electrode being used for cutting and whether fulguration is being performed - this information should be predetermined in each clinic and be available to the LEEP operator when choosing a power setting on the machine for cutting or fulguration. The commonly used power settings for the different loop electrodes are as follows: 1.0 ´ 1.0 cm 30 watts; 1.5 ´ 0.5 cm 35 watts; 2.0 ´ 0.8 cm 40 watts; 2.0 ´ 1.2 cm 50 watts. The power settings for 3 mm and 5 mm ball electrodes are 30 watts and 50 watts, respectively, in the coagulation mode. When possible, a lesion should be removed with one pass of the loop electrode, although this is not always feasible. Four basic operative scenarios are described below:
 table 13.1: The eligibiligy criter...
table 13.1: The eligibiligy criter... table 11.1: Treatment for reproduc...
table 11.1: Treatment for reproduc... figure 13.3: Instrument tray for L...
figure 13.3: Instrument tray for L... figure 4.9: Vaginal speculum cover...
figure 4.9: Vaginal speculum cover...Excision of an ectocervical lesion with one pass (Figures 13.4 and 13.5)
The operator should use a loop that is wider than the lesion(s) and the transformation zone to be removed. The depth of the loop should be at least 5 mm (height from the cross bar to the farthest part of the wire arc). Often one may use a 2.0 ´ 0.8 cm oval loop. To maintain the ideal geometry and depth of cut, it is desirable to orient the surface of the ectocervix at right angles to the handle of the cutting electrode holder - that is, to keep the cross bar parallel to the ectocervix. To begin, local anaesthesia is administered, the electrosurgical generator is set to the appropriate power and blended cutting setting, and the smoke evacuation system is turned on. When the loop is poised just above the starting point, but not touching the cervical surface, the operator activates the current with a foot pedal or finger switch on the electrode holder. The loop is introduced into the tissue 5mm outside the outer boundary of the lesion. It is important not to push the electrode in, but to let it cut its own way; the operator should simply provide directional guidance. The loop is directed gradually into the cervix until the cross bar nearly comes in contact with the epithelial surface. Then the loop is guided along parallel to the surface (horizontally or vertically, depending on the orientation of the direction of cutting) until the point is reached just outside the opposite border of the lesion. The loop is then withdrawn slowly, still keeping it at right angles to the surface. The current is switched off as soon as the loop exits the tissue. It does not matter whether the direction of excision is right to left or vice versa. It also is acceptable to pass the loop from the posterior to the anterior. However, it is not acceptable to pass the loop from the anterior to the posterior, since bleeding or excised tissue curling downward may obscure the visual field.
Once the specimen has been removed and placed in formalin, the setting on the electrosurgical generator is changed to fulguration and the appropriate power is selected. The surface of the excisional crater is fulgurated using 3 or 5 mm ball electrode, in the coagulation mode. The edges of the crater should also be fulgurated to preserve the squamocolumnar junction in the visible ectocervix. If active bleeding occurs and is difficult to control using the ball electrode, a macroneedle style electrode can be effectively used to apply the fulguration current in a much more concentrated (higher current density) and localized fashion to a bleeding site. If satisfactory haemostasis has been obtained, the surface of the crater is then coated with Monsels paste and the speculum is removed. It is a general observation that an extremely nervous patient tends to bleed more than a relaxed one - another good reason to communicate with the patient throughout the procedure and to try to calm her fears.
If bleeding is difficult to stop despite use of the methods outlined above, the base of the excisional crater should be liberally coated with Monsels paste and the vagina packed with gauze. The woman should be asked to wait for several hours before removing the pack. This complication appears to occur more frequently in women with cervicitis.
Once the specimen has been removed and placed in formalin, the setting on the electrosurgical generator is changed to fulguration and the appropriate power is selected. The surface of the excisional crater is fulgurated using 3 or 5 mm ball electrode, in the coagulation mode. The edges of the crater should also be fulgurated to preserve the squamocolumnar junction in the visible ectocervix. If active bleeding occurs and is difficult to control using the ball electrode, a macroneedle style electrode can be effectively used to apply the fulguration current in a much more concentrated (higher current density) and localized fashion to a bleeding site. If satisfactory haemostasis has been obtained, the surface of the crater is then coated with Monsels paste and the speculum is removed. It is a general observation that an extremely nervous patient tends to bleed more than a relaxed one - another good reason to communicate with the patient throughout the procedure and to try to calm her fears.
If bleeding is difficult to stop despite use of the methods outlined above, the base of the excisional crater should be liberally coated with Monsels paste and the vagina packed with gauze. The woman should be asked to wait for several hours before removing the pack. This complication appears to occur more frequently in women with cervicitis.
 figure 13.4: Excision of an ectoce...
figure 13.4: Excision of an ectoce... figure 13.5: Excision of an ectoce...
figure 13.5: Excision of an ectoce...Excision of an ectocervical lesion with multiple passes (Figure 13.6)
If the diameter of a lesion exceeds the width of the largest loop (usually 2 cm), the lesion must be removed with multiple passes using one or more sizes of loop. Using the basic method described above (Figure 13.3), the central part of the lesion usually is removed first. The remaining parts of the lesion in the periphery are then removed by one or more separate passes. All specimens are preserved for pathological examination.
 figure 13.6: Excision of an ectoce...
figure 13.6: Excision of an ectoce... figure 13.3: Instrument tray for L...
figure 13.3: Instrument tray for L...Excision of ectocervical plus endocervical lesions (Figures 13.7 and 13.8)
If a lesion involves the endocervical canal and is not likely to be removed with the depth of the usual single-layer pass as described above and shown in figures 13.4 13.5.jpg&leg=')">and 13.5, a two-layer excisional method can be used. When lesions involve the canal, most of them extend for a linear length of 1 cm or less into the endocervical canal. Older women and women with CIN 3 are likely to have longer lesions and require a second layer - composed wholly of the endocervical canal - to be excised.
Usually the ectocervical portion of this type of lesion that extends into the canal can be excised by one pass of a large oval (2.0 ´ 0.8 cm) loop. The remaining tissue in the endocervical canal can be excised using a smaller loop - usually a square loop with a 1.0 ´ 1.0 cm configuration - but care must be taken not to go any deeper than is necessary to completely excise the lesion and a margin of normal tissue. This type of excision can reach a maximum of 1.6 cm into the endocervical canal (13.7.jpg&leg=')">Figure 13.7). Excision of this depth should be attempted only when absolutely necessary, due to increased risk of bleeding and stenosis as the depth of excision increases. LEEP should not be used if the distal or upper extent of the lesion in the canal cannot be seen and if the distal end of the lesion extends more than 1 cm into the canal. Such patients should undergo cold-knife conization. Since this two-step method requires adequate performance of the basic LEEP procedure, it is recommended that it should not be attempted until the operator is comfortable and competent with the basic LEEP. Women with lesions that extend further up into the canal need cold-knife conization to properly assess the endocervical canal. Lesions with vaginal extension If the lesion extends onto the vagina, it is preferable to use the ball electrode for electrofulguration on the peripheral, vaginal part of the lesion and LEEP on the central, cervical part of the lesion. The treatment of these vaginal lesions is beyond the scope of this manual and the LEEP treatment referred to here deals only with the type of lesions shown in Figures 13.4 and 13.6, 13.7, 13.8 and described above. Interested readers may refer to standard text books ( Wright et al., 1992; Wright et al., 19.5).
Usually the ectocervical portion of this type of lesion that extends into the canal can be excised by one pass of a large oval (2.0 ´ 0.8 cm) loop. The remaining tissue in the endocervical canal can be excised using a smaller loop - usually a square loop with a 1.0 ´ 1.0 cm configuration - but care must be taken not to go any deeper than is necessary to completely excise the lesion and a margin of normal tissue. This type of excision can reach a maximum of 1.6 cm into the endocervical canal (13.7.jpg&leg=')">Figure 13.7). Excision of this depth should be attempted only when absolutely necessary, due to increased risk of bleeding and stenosis as the depth of excision increases. LEEP should not be used if the distal or upper extent of the lesion in the canal cannot be seen and if the distal end of the lesion extends more than 1 cm into the canal. Such patients should undergo cold-knife conization. Since this two-step method requires adequate performance of the basic LEEP procedure, it is recommended that it should not be attempted until the operator is comfortable and competent with the basic LEEP. Women with lesions that extend further up into the canal need cold-knife conization to properly assess the endocervical canal. Lesions with vaginal extension If the lesion extends onto the vagina, it is preferable to use the ball electrode for electrofulguration on the peripheral, vaginal part of the lesion and LEEP on the central, cervical part of the lesion. The treatment of these vaginal lesions is beyond the scope of this manual and the LEEP treatment referred to here deals only with the type of lesions shown in Figures 13.4 and 13.6, 13.7, 13.8 and described above. Interested readers may refer to standard text books ( Wright et al., 1992; Wright et al., 19.5).
 figure 13.7: Excision of ectocervi...
figure 13.7: Excision of ectocervi... figure 13.8: Excision of an ectoce...
figure 13.8: Excision of an ectoce... figure 13.4: Excision of an ectoce...
figure 13.4: Excision of an ectoce... figure 13.5: Excision of an ectoce...
figure 13.5: Excision of an ectoce... figure 13.6: Excision of an ectoce...
figure 13.6: Excision of an ectoce... FIGURE 13.8: Excision of an ectoce...
FIGURE 13.8: Excision of an ectoce...Follow-up care after LEEP
Women should receive instructions on self-care and what symptoms to expect after treatment. If appropriate, written instructions should be provided. Women should be advised that they will have a brown or black discharge lasting between a few days and two weeks. They should be advised to promptly report back if the discharge persists for more than two weeks, if discharge becomes malodorous and/or is associated with lower abdominal pain or if profuse bleeding develops. Women should be advised not to use a vaginal douche or tampons, or to have sexual intercourse for one month. The appearance of cervix three months and one year after LEEP is shown in 13.9.jpg&leg=')">Figures 13.9 and 13.10.
The effect of LEEP treatment on the potential transmissibility of HIV (to or from women) during the healing phase is not known. HIV-1 shedding in the vaginal secretions after treatment of CIN in HIV-positive women has been demonstrated ( Wright et al., 2001). Therefore, the authors suggest advising all women that LEEP treatment may increase the transmissibility of HIV and that using condoms is an effective means of prevention. Condoms should be used for period of 6-8 weeks. Ideally, a supply of condoms should be available, free of charge, at colposcopy clinics in settings where HIV infection is endemic.
A follow-up appointment should be made for review at 9-12 months after treatment; the management plan should follow the scheme outlined in Chapter 11. Management of women who have persistent disease at the follow-up visit(s) is discussed in the next section.
The effect of LEEP treatment on the potential transmissibility of HIV (to or from women) during the healing phase is not known. HIV-1 shedding in the vaginal secretions after treatment of CIN in HIV-positive women has been demonstrated ( Wright et al., 2001). Therefore, the authors suggest advising all women that LEEP treatment may increase the transmissibility of HIV and that using condoms is an effective means of prevention. Condoms should be used for period of 6-8 weeks. Ideally, a supply of condoms should be available, free of charge, at colposcopy clinics in settings where HIV infection is endemic.
A follow-up appointment should be made for review at 9-12 months after treatment; the management plan should follow the scheme outlined in Chapter 11. Management of women who have persistent disease at the follow-up visit(s) is discussed in the next section.
 figure 13.9: Appearance of the cer...
figure 13.9: Appearance of the cer... figure 13.10: Appearance of the ce...
figure 13.10: Appearance of the ce...Adverse effects, complications, and long-term sequelae of LEEP
Most women experience some transitory pain from the injection of local anaesthetic into the cervix. Severe perioperative bleeding occurs after 2% or less of LEEP procedures. Women should be advised to contact the clinic if they have any concerns during the post-operative period. It is advisable to give written post-operative instructions that outline the following points. Few women complain of post-operative pain. If post-operative pain occurs, it usually is similar to cramps; women should be instructed to use oral analgesics such as acetaminophen or ibuprofen, if necessary. A blood-tinged, dark brown (from the Monsels paste) mucus discharge usually lasts for one or two weeks after treatment. Severe and moderate post-operative bleeding occurs in a few women, who should be seen promptly. Healing after LEEP usually takes place within a month.
When post-operative bleeding occurs, it usually appears 4-6 days after treatment and often from the posterior lip of cervix. This bleeding can usually be controlled by fulguration, applying Monsels paste, or using a silver nitrate applicator stick. Rarely, placement of a suture at the bleeding site is necessary. The risk of post-operative infection is very small and can probably be reduced even more by delaying surgical treatment until any woman with a likely diagnosis of PID, cervicitis, vaginal trichomoniasis or bacterial vaginosis has been adequately treated and recovered. If a woman presents post-operatively with a malodorous discharge, it should be cultured if possible and empirical treatment prescribed with antibiotics that are effective for PID (see Table 11.1). In developing countries, it may be preferable to institute routine presumptive treatment with antibiotics after LEEP (doxycycline 100 mg orally, two times a day, for seven days and metronidazole 400 mg orally, three times a day, for seven days).
The squamocolumnar junction is in the endocervical canal at the follow-up evaluation in approximately 2% of women. This presents a challenge for adequate colposcopic examination and cytological sampling. Women should be warned that cervical stenosis, partial or complete, is rarely encountered (probably less than 1%), but is more common in menopausal women.
When post-operative bleeding occurs, it usually appears 4-6 days after treatment and often from the posterior lip of cervix. This bleeding can usually be controlled by fulguration, applying Monsels paste, or using a silver nitrate applicator stick. Rarely, placement of a suture at the bleeding site is necessary. The risk of post-operative infection is very small and can probably be reduced even more by delaying surgical treatment until any woman with a likely diagnosis of PID, cervicitis, vaginal trichomoniasis or bacterial vaginosis has been adequately treated and recovered. If a woman presents post-operatively with a malodorous discharge, it should be cultured if possible and empirical treatment prescribed with antibiotics that are effective for PID (see Table 11.1). In developing countries, it may be preferable to institute routine presumptive treatment with antibiotics after LEEP (doxycycline 100 mg orally, two times a day, for seven days and metronidazole 400 mg orally, three times a day, for seven days).
The squamocolumnar junction is in the endocervical canal at the follow-up evaluation in approximately 2% of women. This presents a challenge for adequate colposcopic examination and cytological sampling. Women should be warned that cervical stenosis, partial or complete, is rarely encountered (probably less than 1%), but is more common in menopausal women.
 table 11.1: Treatment for reproduc...
table 11.1: Treatment for reproduc...Management of women with persistent lesions at follow-up
All women, regardless of whether or not the pathology report states that the excisional margins are clear, should be followed up at 9 - 12 months from treatment to evaluate regression or persistence of lesions and complications. Treatment failures (persistent lesion(s) at follow-up) are detected in less than 10% of women when they are checked at the follow-up appointment. It is advisable to biopsy all persistent lesions to rule out the presence of unsuspected invasive carcinoma. Persistent lesions should be re-treated with cryotherapy or LEEP or cold- knife conization, as appropriate.



